Initial microfluidic devices were fabricated using methods from the microelectronics industry, but cleanrooms, expensive process instrumentation and materials severely restricted access to the technology, especially for chemists and bioscientists, who were not traditional users of cleanrooms. The Whitesides group dramatically changed the field of microfluidics by introducing PDMS-based microfluidics.4 This simple, robust route to replica mold and seal low-cost elastomeric chips gained widespread popularity in academic communities, unlocking the creativity of researchers to craft their own devices for diverse purposes ranging from studies of fundamental physics, to medical diagnostics.
Paper and thread-based microfluidics
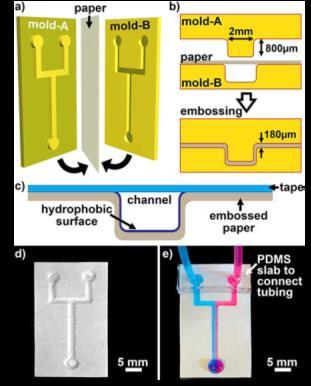
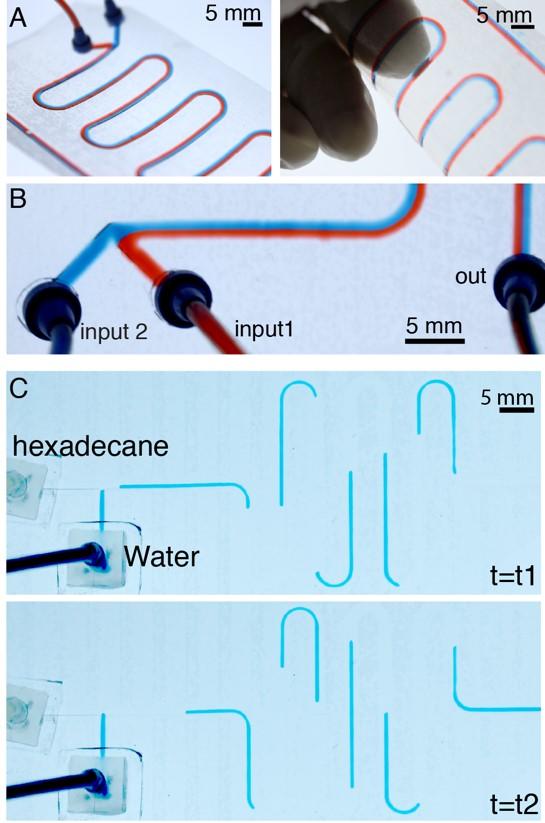
We have sought to develop even simpler and lower-cost microfluidic technologies based on materials such as paper and threads.1,3 One class of paper devices is powered by natural occurring wicking, when water wets the hydrophilic cellulose fibers. We have developed a simple method to create fluidic circuits in paper by using a conventional office printer that produces a hydrophobic barrier to selectively block wicking in areas covered with wax ink.2 Stacking these individual paper sheets into multilayered devices can make even more versatile three-dimensional liquid handling systems (Figure 1).4, 6 We have extended the liquid handling possibilities of these systems, and incorporated electrodes (Figure 2) and electronics onto the paper substrates and to perform low-cost diagnostic assays needed in the developing World.7 Similar to paper, cotton threads are ideal at wicking liquids and acting as one-dimensional fluidic channels (Figure 3).2 We have also used hydrophobic papers to construct flow channels by cutting, engraving (Figure 3) and embossing (Figure 4).8 These channels have an open cavity and require external pressure to drive the flow. We have demonstrated, that these channels can be used for multiphase droplet microfluidics, they support gas exchange through porous paper, and the flows can be adjusted by foldable valves. The group has also demonstrated use of other ubiquitous materials such as cellophane for developing microfluidics with complex liquid handling and analysis capability (Figure 6).
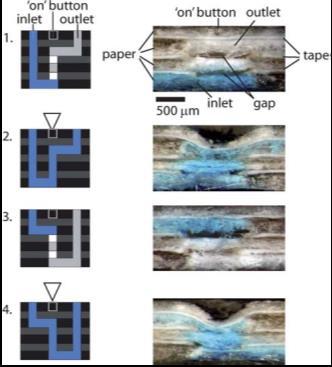
|
Figure 1. Programmable diagnostic device made from paper and tape. This device is fabricated by stacking multiple layers of paper, having fluidic connectivity both in, and through, the plane of the paper sheet. If a gap is made in some of the layers, vertical connectivity is lost until device is squeezed (for example with pencil tip). This closes the gap and connects the flow path. This principle allows us to make very simple, single use mechanical push-button valves for paper microfluidic devices.6 |
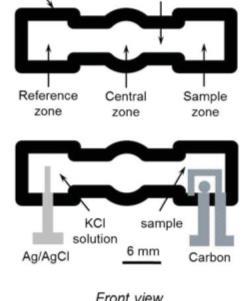
|
Figure 2. Three electrode electrochemical paper analytical device, including carbon based working and counter electrodes and stable Ag/AgCl reference electrode.13 |
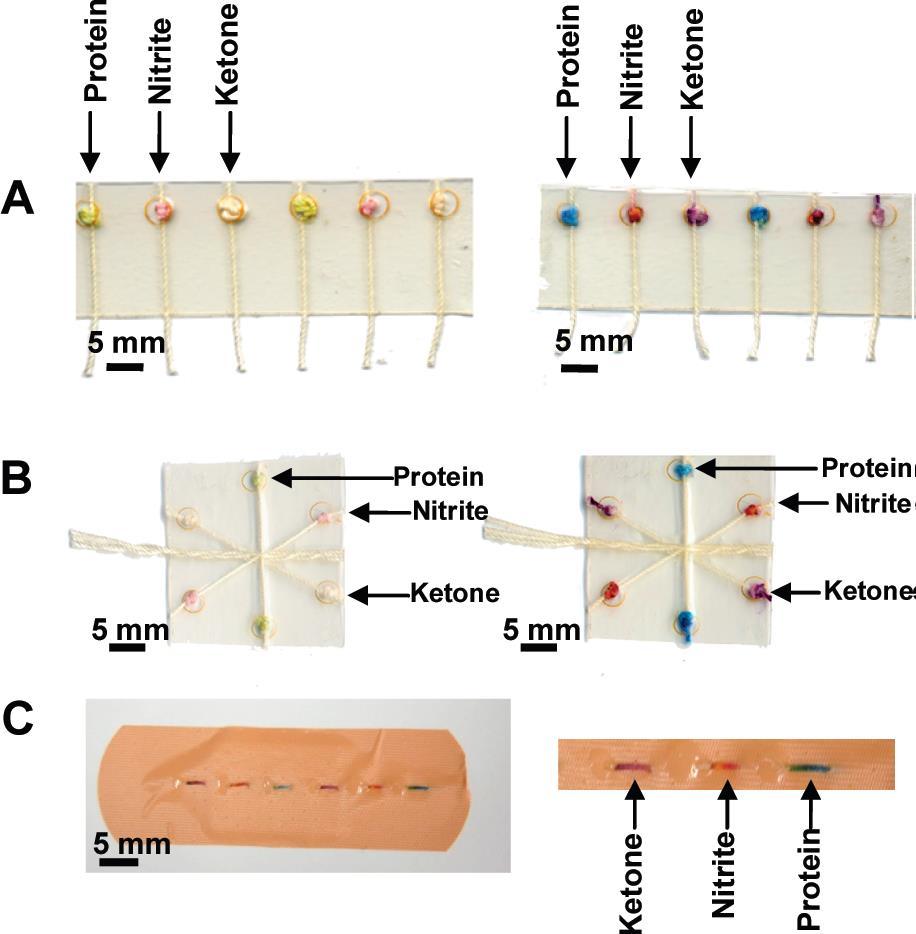
|
Figure 3. Colorimetric assays performed using the (A) woven array device, (B) branching device, (C) sewn array design.1 |
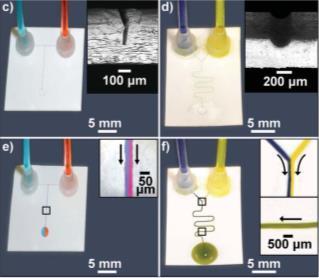
|
Figure 4. Open channel microfluidic devices fabricated in omniphobic paper by cutting (c, e) and engraving (d, f). |
|
Figure 5. Fabrication of low-cost open channel paper-based microfluidic devices by embossing. |
|
Figure 6. Photographs of the Y-junction microfluidic device, fabricated with nitrocellulose-coated cellophane (CNS), displaying a laminar flow created by pumping red (input 2) and blue (input 1) aqueous dyes at a speed of 50 µL/min. Photographs of multiphase flow using the T-junction cellophane microfluidic device, showing the generation of slugs in a hexadecane / water system (both solutions had a flow rate of 30 µL/min). |
Paper microfluidics for bioanalysis
The Whitesides lab has explored application of 3D microfluidics as new tools for bioanalysis, specifically for studying cell cultures (Figure 7).9-12 Cell cultures in stacked, paper-supported gels offer a flexible approach to study cell responses against 3D molecular gradients of oxygen, nutrients, chemicals, or biological molecules. This approach can be useful to mimic tissue- and organ-level functions in vitro and in vivo. Permeating a suspension of cells in hydrogels into the paper matrix creates thin 3D tissues in which the growth of cells is not limited by mass transport of oxygen or nutrients. Stacking and de-stacking layers of paper impregnated with suspensions of cells in hydrogels make it possible to control oxygen and nutrient gradients in three dimensions (3D), and to analyze molecular and genetic responses. Stacking assembles the tissue construct whereas de-stacking disassembles it, and permits its analysis. Using these systems, we study the role of molecular gradients on drug and radiation sensitivity of cancer cells in 3D tumors, high-throughput testing of the effect of soluble compounds, cardiac ischemia-reperfusion injury, mineralization in 3D constructs, and the formation and control of bacterial communities (biofilms) in 3D.9-13
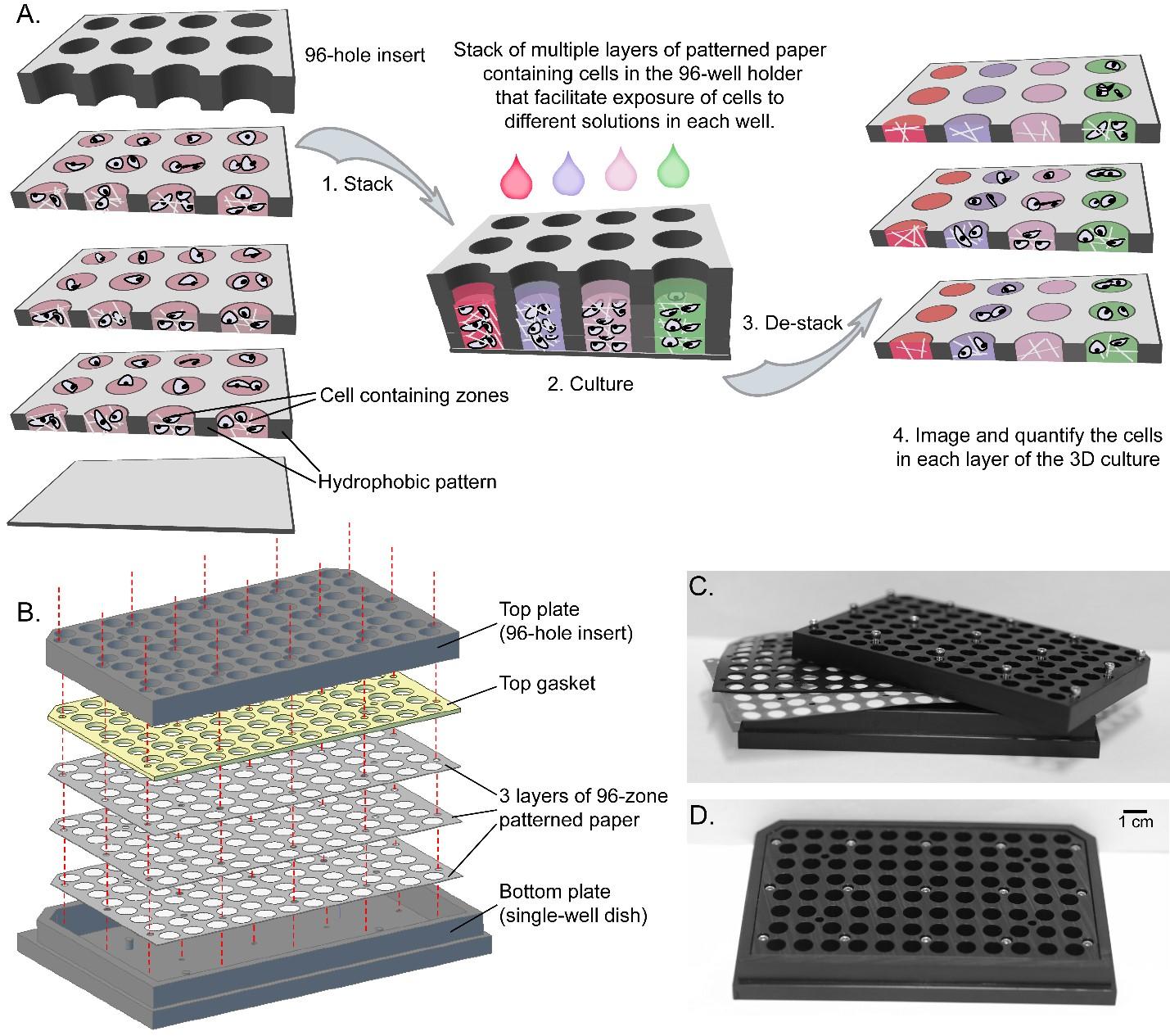
Figure 7. General Design of the 96-well Holder Containing Multiple Sheets of Paper Impregnated with Mammalian Cells.12
References:
